Light Solution For A Deadly Problem
Editorial / March 18, 2026
A flash of blue light may help solve one of the most persistent water pollution problems. For years, scientists have been grappling with a chemical built to evade modern water treatment systems. The compound, known as 1,4-dioxane, is commonly used in a range of industrial processes, from pharmaceutical production to the manufacture of polymers, cosmetics and cleaning products. Over time, its widespread use has allowed it to seep into groundwater and wastewater systems worldwide.
The compound, classified as a probable human carcinogen, may damage the liver and kidneys. To make things worse, the chemical dissolves in water and does not evaporate quickly. It can also move through groundwater systems with ease. As a result, traces of the compound have been detected in drinking water sources in several countries, raising questions about the risks of long-term exposure through everyday consumption.
Part of the challenge lies in the compound's stubborn chemistry. Biological treatments, filtration systems, and chemical processes often fail to break down the pollutant effectively. This has made 1,4-dioxane an increasingly urgent concern.
The most common solution has been to rely on advanced oxidation processes, which use ultraviolet light along with powerful oxidising chemicals to break down pollutants. While such techniques can be effective, they are often energy-intensive and require specialised infrastructure. In some cases, they can also produce additional chemical by-products that must be treated separately, adding another layer of complexity.
Faced with these limitations, researchers have begun exploring alternative approaches. A team of scientists believe the solution may lie in a different way of thinking about the problem. Instead of trying to destroy the pollutant, their idea is to modify its chemical structure so it becomes easier to remove from water.
The battle against 1,4-dioxane brought together Dr V M Rajesh, an associate professor in the Department of Chemical Engineering, and Professor Subhabrata Sen from the Department of Chemistry, along with Professor Ludovic Gremaud from the University of Applied Sciences and Arts of Western Switzerland. They were assisted by doctoral researchers Haya Khan and Shreekanth Murugan.
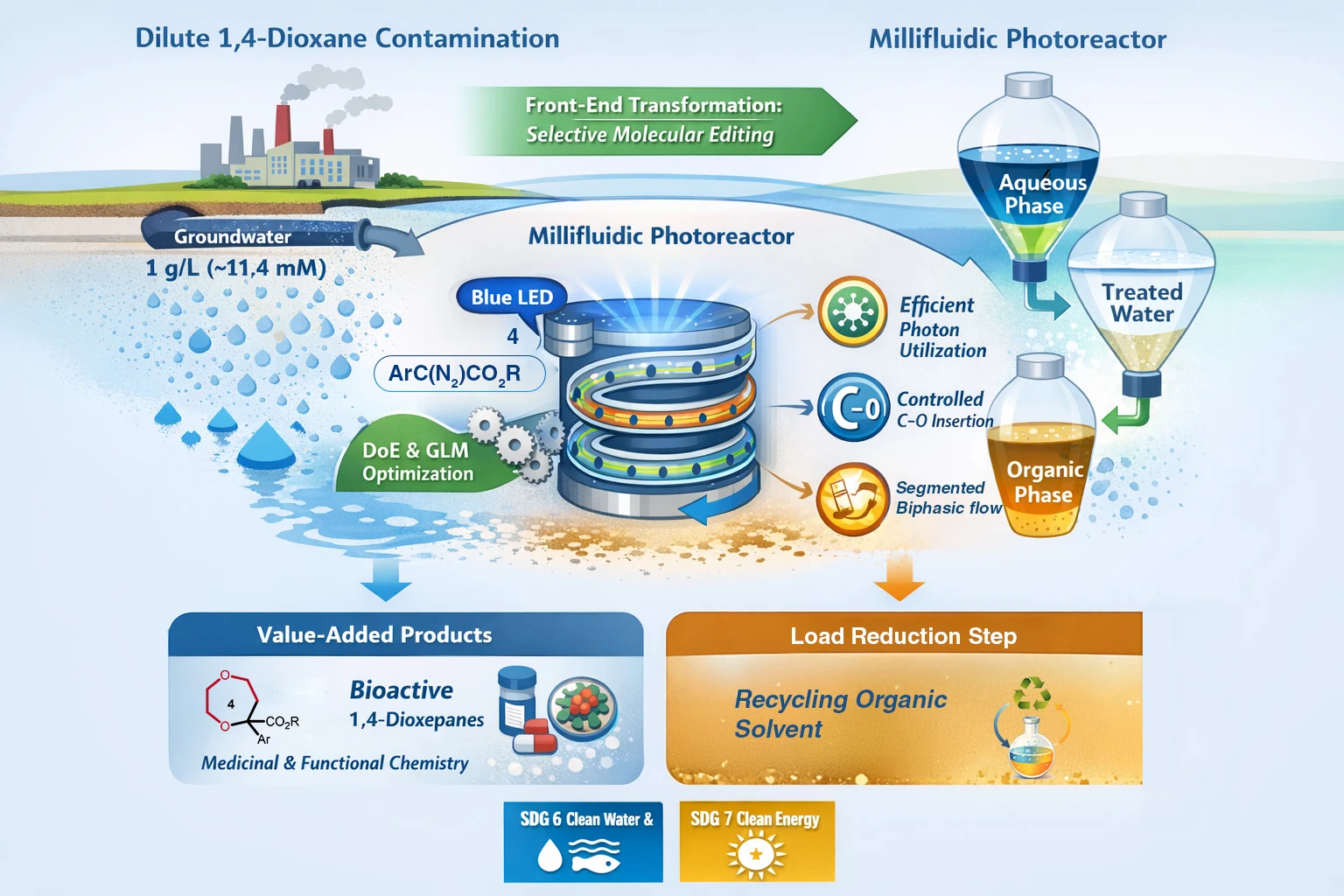
Drawing deep into a concept in organic chemistry known as skeletal editing, they developed a technique that reshapes a molecule by rearranging the bonds that hold its atoms together. In their experiments, they used blue LED light to activate a group of chemical reagents called aryl diazoacetates, which then interact with the 1,4-dioxane molecule and begin to alter its structure. Once activated by light, these reagents form highly reactive intermediates that interact with the 1,4-dioxane molecule.
The reaction opens the ring structure of the pollutant, forming a related compound, 1,4-dioxepane. Although the change in structure is relatively small, it has an important consequence. The new compound does not dissolve in water as readily as the original pollutant, which means it can be more readily separated from the water stream during treatment.
To efficiently carry out the reaction, the researchers developed a continuous-flow millifluidic photoreactor. Instead of allowing chemicals to react in a single container, the system channels liquids through narrow pathways only a few millimetres wide. Inside the reactor, contaminated water flows alongside an organic solvent, forming tiny droplets as the liquids move through the device.
As the mixture passes through the reactor, blue LED light illuminates the flowing droplets and triggers the skeletal editing reaction. The narrow channels help ensure that the liquids mix effectively and that light reaches the reacting molecules. The droplet structure also increases the surface area where reactions occur, allowing the transformation to proceed more quickly and efficiently.
Laboratory tests have produced promising results. Under optimised conditions, the system converted more than 93 per cent of the 1,4-dioxane into dioxepane derivatives. Because the newly formed molecules are more hydrophobic, they tend to move out of the water phase and into the organic solvent stream, where they can be separated and removed.
As water resources worldwide face growing pressure from industrial activity, population growth, and climate change, solutions that balance efficiency with practicality are becoming increasingly important. Sometimes the key to dealing with a stubborn pollutant lies not in overwhelming it with chemistry, but in subtly reshaping its molecular structure. In this case, that transformation begins with a small but powerful ingredient: a beam of blue light.
More Blogs

The Hawthornden Literary Retreat bestowed on Dr Sambudha Sen to complete the manuscript of a novel
Professor Sambudha Sen, Head of the Department of English at Shiv Nadar Institution of Eminence, Delhi-NCR, was awarded a residency at the...

The Power of the Moving Body
Movement is an innate bodily action that humans have been exhibiting for the longest time. Long before language was invented, the body was the...

How Does A Multi-Disciplinary Approach To Education Enhance Learning And Prepare Students For A Multi-Faceted World?
In today’s world, where businesses are changing almost every day, it is the responsibility of educational institutes to provide holistic...